---teachoo-01.jpg)
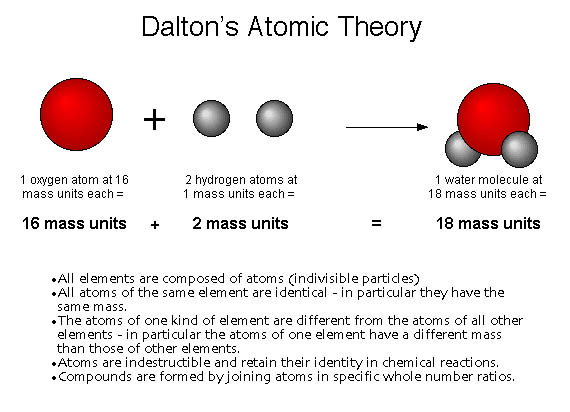
: Dalton's assumptions Five steps forward and one step back. Next, we'll see how Dalton's postulates can be used to estimate atomic weights from experimental data, and how they explain three basic laws of chemistry. As a result, Dalton's atomic weights for oxygen and nitrogen were incorrect and his experimental data did not support many of the conclusions he drew from it.Ī consistent set of atomic weights was absolutely essential before the theory could be accepted and applied. He had no experimental evidence to support this postulate, and it lead him to mistakenly assume that the formula of water was OH and the formula of ammonia was NH. When atoms combine in only one ratio, Dalton said, ".it must be presumed to be a binary one, unless some cause appear to the contrary". Unfortunately, Dalton included an additional postulate that prevented his theory from being accepted for many years. Even Dalton's critics were impressed by the power and simplicity of his explanation, and it persuaded many of them that his atomic theory was worthy of further investigation. When elements react, their atoms sometimes combine in more than one simple, whole-number ratio.ĭalton used this postulate to explain why the weight ratios of nitrogen to oxygen in various nitrogen oxides were themselves simple multiples of each other.Weights could then be used to explain the fixed mass percentages of elementsīy suggesting that compounds contained characteristic atom-to-atom ratios, Dalton effectively explained the law of definite proportions. This postulate suggested a practical strategy for determining relative atomic weights from elemental percentages in compounds. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
